News & Events

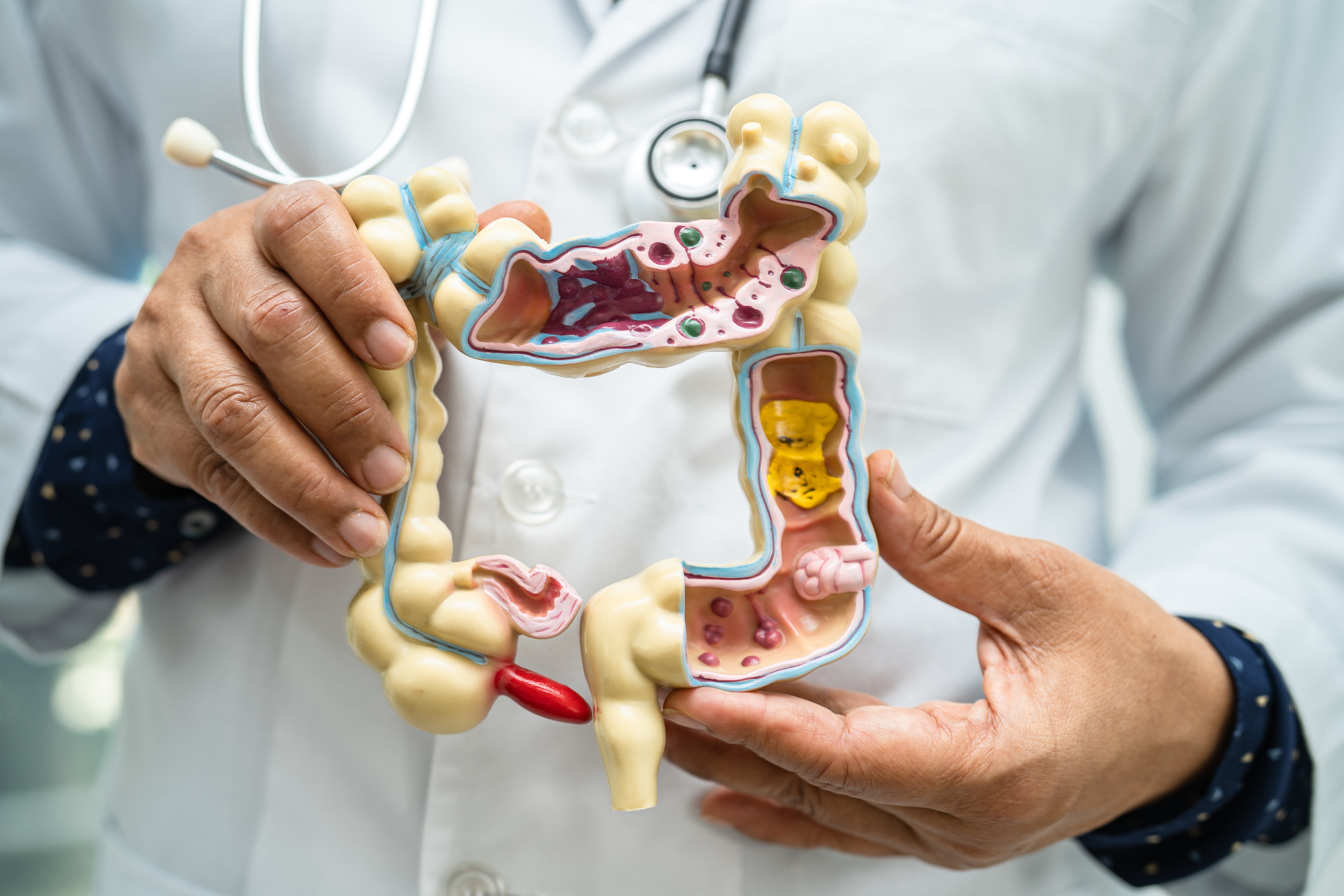
Types of ARM Equipment: Conventional vs. High-Resolution Manometry
It’s vital that clinicians stay informed about advancements in ARM technology to optimize patient care. The rapid advancements of ARM equipment offers new opportunities for more accurate diagnoses and tailored treatments for patients with anorectal disorders. By embracing these advancements, clinicians can enhance their diagnostic capabilities and improve patient outcomes.
Read More
Qualifying Patients for Anorectal Manometry
Anorectal Manometry presents an opportunity for clinics and health systems to enhance their practice. Here’s what you need to know about qualifying patients.
Read More
Increase Profits and Patient Experience with Anorectal Manometry
Anorectal manometry can provide generous reimbursements that increase your profit, while also improving your patients’ experience.
Read More
Is Investing in Anorectal Manometry Worth the Cost?
Discover how the mcompass® system allows you to add anorectal manometry to your practice with a lower investment and a rapid ROI.
Read More
How to Add Pelvic Floor Biofeedback Therapy to Your Practice
Pelvic floor biofeedback therapy can be life-changing for your patients. For your practice, the benefits can be substantive as well.
Read More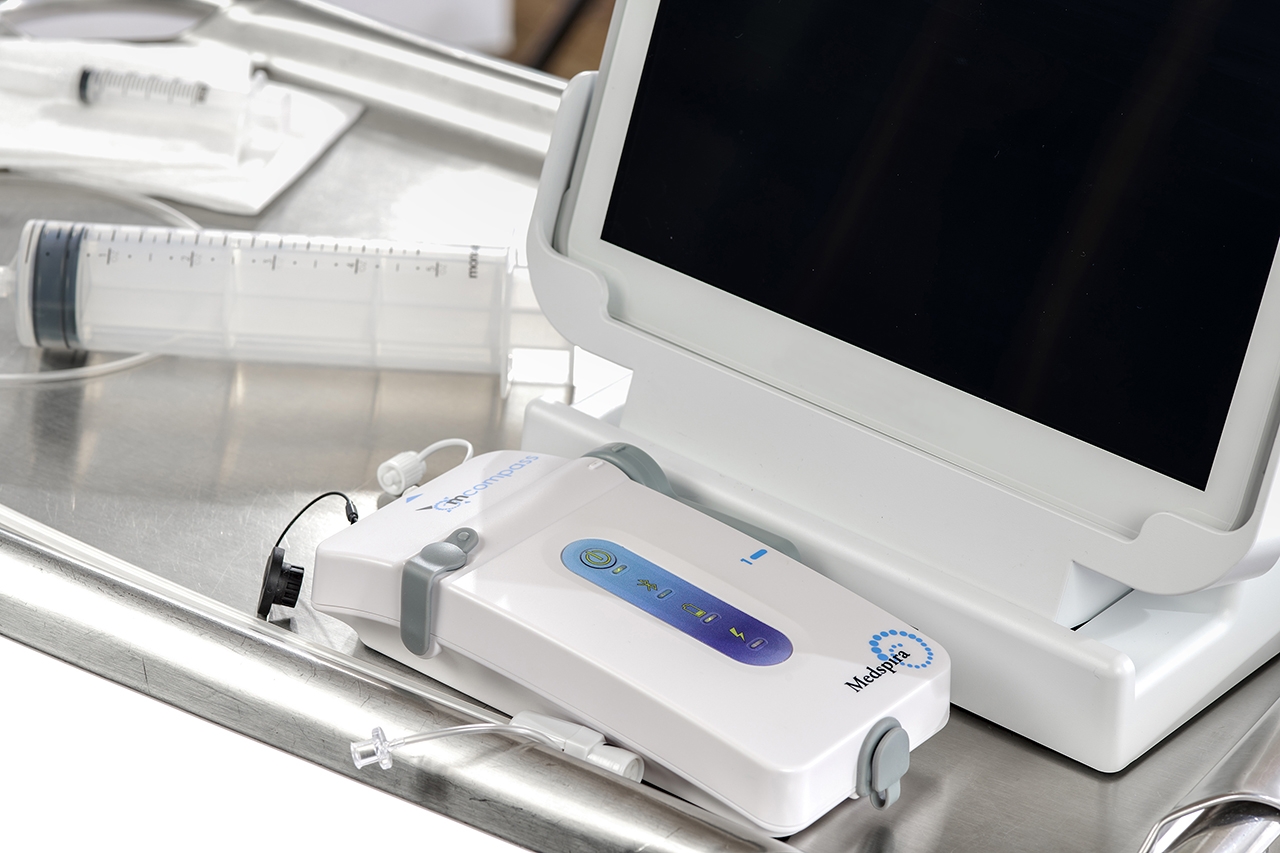
5 Benefits of Portable Anorectal Manometry
Bring anorectal manometry into your office with the portable mcompass system.
Read More
How to Tailor Anorectal Manometry Tests to Pediatric Patients
It’s in the best interest of both the child and the provider to ensure a comfortable and calm anorectal manometry testing environment. Here’s how.
Read More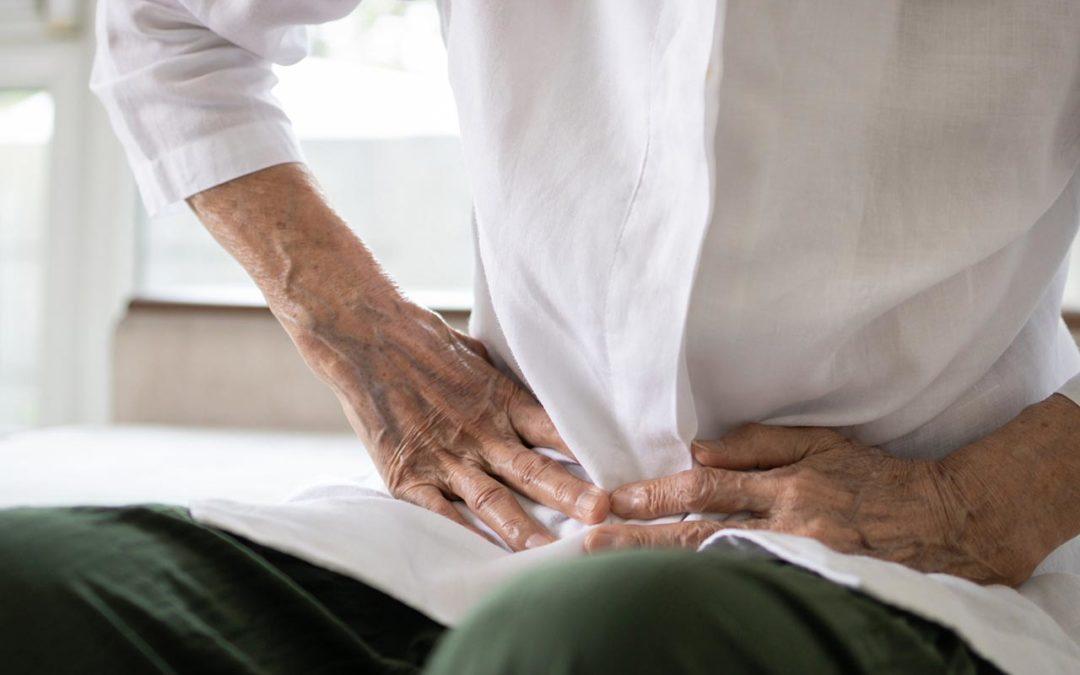
"I Can’t Go" – Explaining Muscle Control and Chronic Constipation
Talking to patients about fecal incontinence was the subject of our last blog and here we continue with the related issue of chronic constipation. Many patients will assume constipation is primarily caused by diet and poor toileting habits.
Read More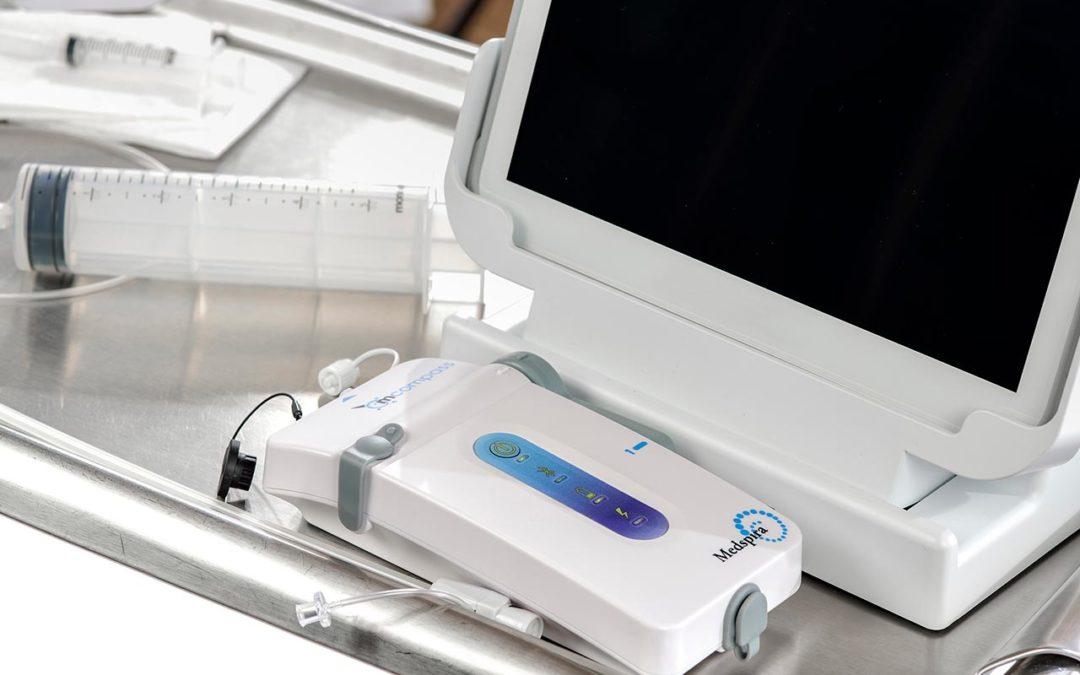
Trailblazing Anorectal Manometry
Imagine elevating patient care, creating an entirely new treatment path and tapping into a lucrative revenue stream simply by incorporating a single, affordable medical device into your practice.
Read More